1 min read
Yposkesi Implements LabWare’s Enterprise Laboratory Platform
Photo: via Yposkesi ALTRINCHAM, UK-- LabWare Limited Yposkesi, an SK pharmteco company, is one of Europe’s largest Contract Development and...
3 min read
LabWare Updated on June 27, 2025

A recent New York Times article highlighted the manufacturing, testing, and distribution processes for the Pfizer-BioNTech Covid-19 messenger RNA (mRNA) based vaccine.
The careful execution, integration, and optimization of multiple steps are critical to the vaccine's safety, effectiveness, and efficient delivery. Pfizer's multifaceted vaccine process takes 60 days from start to finish across numerous locations involving thousands of employees. More than half of this effort goes to quality testing, which must adhere to vaccine manufacturing regulatory requirements, including compliance with current Good Manufacturing Practices (cGMP).
The production of modern vaccines at Pfizer and other major manufacturers relies heavily on tried-and-true LabWare® LIMS and LabWare ELN to manage enormous amounts of information created during the complex processes in a vaccine manufacturing campaign. Their analytical research and development (R&D) and quality assurance and quality control (QAQC) laboratories generate critical data for decision-making about vaccine efficacy and safety. Accurate vaccine sample details, storage documentation, test assignment, calculations, and reporting count on LabWare LIMS and ELN to deliver automation and data integrity in vaccine manufacturing that is impossible with manual processes.
Let's look at the vaccine manufacturing steps where LabWare plays a vital role in successful vaccine production and delivery.
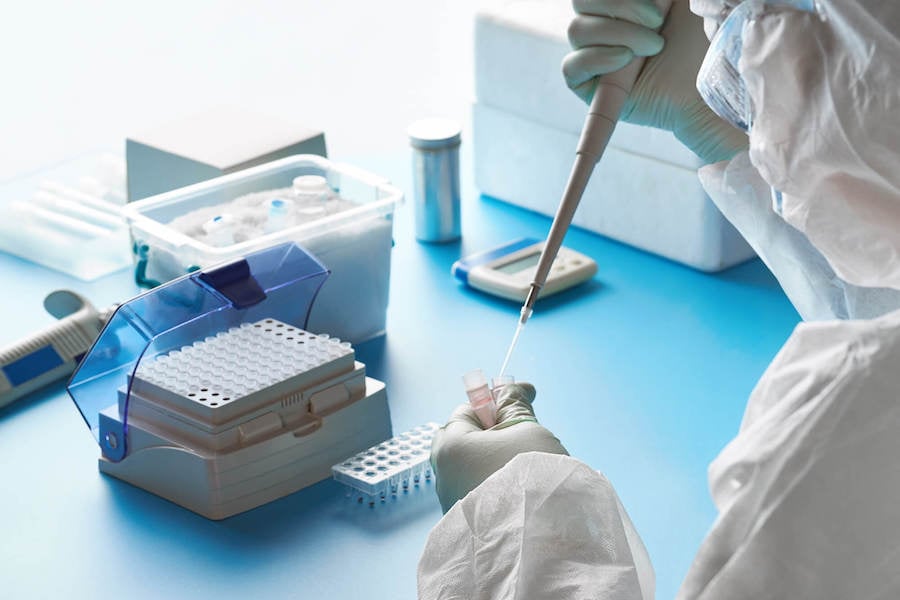
A single vial of DNA plasmids could be the source of millions of doses of the Covid-19 vaccine. Many vials must be stored in a carefully controlled environment and tracked entirely from the beginning to the end of their lifecycle. Robust storage location, specimen tracking, and logistics capabilities with flexible searching, specimen testing, and workflow management features in LabWare LIMS are ideal for controlling this precious starting material for the vaccine.
LabWare tracks the location and condition of plasmid vials and monitors any work that has begun on them. Vial movements from one place to another are automatically tracked in LabWare, making traceability and documentation smooth and straightforward. Barcoding the vial labels automates information gathering and processing, ensuring sampling events are fully traceable with a close eye kept on a strict specimen chain of custody.
Modified batches of bacteria cells act as "factories" for growing large volumes of the Covid-19 vaccine plasmids. Because vaccine manufacturing uses these biological materials in sterile and warm environments, potential contamination with unintended microorganisms could compromise finished vaccine viability and safety. LabWare LIMS can provide the necessary planning, scheduling, sampling, test assignment, and reporting to detect and avoid corruption of the vaccine at all research and production stages.
Protocols enforced by LabWare LIMS ensure vaccine scientists and technicians follow guidelines precisely to minimize contamination during their work. LIMS streamlines and controls the processes and large volumes of data from the extensive tracking and testing of the bacteria cell lines, DNA plasmids, and mRNA at all steps in the vaccine process. Ensuring the integrity of vaccine manufacturing facilities is further enhanced by the addition of LabWare ELN to readily create protocol templates for methods such as sterility and air particulate monitoring.
Manufacturers must consistently and diligently monitor the quality of their vaccines throughout the entire process. LabWare has proven to be a core solution for vaccine QAQC testing. Testing results need to be associated with each production batch, and relevant documentation must be readily available. LabWare LIMS handles the testing of the vaccine plasmids for purity, including the automatic triggering of investigations when out-of-specification results occur.
The results of this quality testing step are crucial in determining if the circular plasmids can move forward to the linearization step, which separates the coronavirus genes into straight segments. The purified DNA sequences are tested again using LabWare to manage the data and specifications and verify the performance and content at each step.
The completed Covid-19 vaccine requires processing the DNA sequences into messenger RNA (mRNA), the active ingredient of the Pfizer-BioNTech vaccine. Analytical scientists rely on LabWare LIMS and ELN to manage the data during repeated testing of the mRNA to verify its purity and confirm the genetic sequence is correct. After weeks of rigorous testing, data collection, and review in LabWare, the vaccine will be ready to ship.
The urgency to deliver safe and effective Covid-19 vaccines at record speeds puts pressure on manufacturers to provide rapid and accurate reporting of complex testing results. Documentation and batch records are submitted and reviewed by government regulators. The regulators authorize the release of vaccines for distribution. LabWare provides the precise graphical and data representations needed to align with a vaccine quality program's exact and unique needs.
LabWare LIMS tracks and analyzes historical vaccine testing data, which can identify trends over time. If there are any recurring issues involving samples, having access to these time trends can help pinpoint the source of problems. Deviations can arise from supplier issues, human error, instrument malfunctions, or inconsistencies in hardware. Tracking data over time in LabWare improves quality and allows access to trends that can anticipate future issues. QC lab managers can respond by taking more holistic corrective actions to influence laboratory quality positively.
Key Performance Indicators (KPIs) are invaluable tools that measure vaccine manufacturing effectiveness. LabWare KPIs help vaccine testing laboratories stay accountable to regulatory requirements and efficient processes. Every laboratory can configure KPIs to match their unique needs. These indicators provide real-time operational metrics at any time. Easy-to-digest dashboards present KPIs that the laboratory can reference at any time and make it simple to obtain and process relevant information.
The remarkable achievements of Pfizer and other major manufacturers in rapidly delivering new safe and effective vaccines to address the Covid-19 pandemic rely on underlying quality programs and systems. Implementing a LIMS for vaccine development laboratories and manufacturing facilities simplifies data management, increases automation, and optimizes workflows. These efficiencies further streamline the vaccine production process, ultimately allowing companies to scale up their operations efficiently.
LabWare LIMS and ELN have been leading mission-critical components to vaccine development and QAQC testing for years and continue to help companies, like Pfizer rapidly meet the demand for new vaccines.

1 min read
Photo: via Yposkesi ALTRINCHAM, UK-- LabWare Limited Yposkesi, an SK pharmteco company, is one of Europe’s largest Contract Development and...

1 min read
Contract Development and Manufacturing Organizations (CDMOs) lose millions annually due to manufacturing delays, client changeovers, and compliance...

1 min read
Note: This story was featured in the April 2021 edition of LABInsights and has been edited for clarity. Denkavit recently expanded the functionality...