2 min read
LabWare Nordic User Meeting 2025: A Gathering of Innovation and Insight
LabWare is glad to announce its upcoming Nordic User Meeting, scheduled for 26–27 November 2025 at the Clarion...
Add text here
Add text here







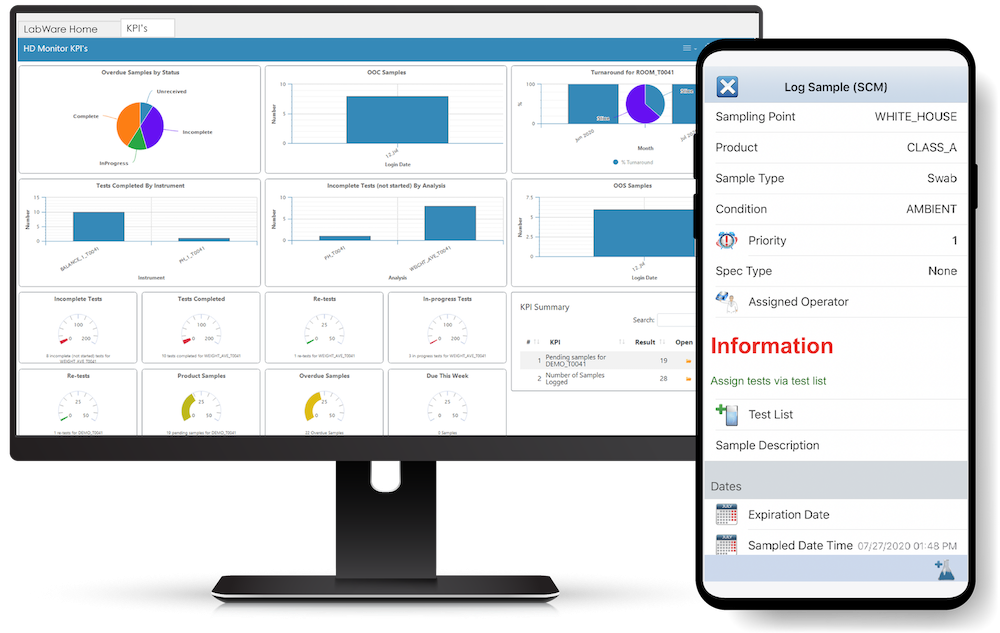
LabWare supports laboratory automation and paperless operations with native tools that simplify the task of interfacing with a variety of laboratory analyzers and robotics platforms as well as a configurable rules engine that can be used to enable, expedite, and increase the efficiency and effectiveness of the laboratory operation.

LabWare's integrated electronic laboratory notebook provides additional capability to manage large data sets in plate based testing and flexibility to document research experiments.

LabWare's LIS enables fully integrated diagnostic reporting with the flexibility to manage the diverse needs across a variety of pathology laboratory departments including Chemistry, Hematology, Urinalysis, Coagulation, Virology, Immunology, Serology, Microbiology, Molecular Biology, Cytogenetics, Flow Cytometry, Anatomic Pathology, Cytology, and Toxicology.

LabWare MOBILE (available on Android and iOS devices) allows users to easily connect to their LabWare systems, see their data, and perform customer-defined tasks. Learn More >
LabWare's flexible workflow tools automate clinical test order processing through the entire order life cycle.
LabWare’s configurable order management, specimen collection, and accessioning tools address various situations, including manual order entry, CPOE via an automated interface with HIS or EMR, interface with another LIS, and import of batch order files. Other integrated features include sample collection/phlebotomy, built-in tools to capture ICD-9/10 codes with medical necessity checking, recurring orders, cloning and merging orders, double data entry, question forms, and label printing.
Use quality management tools, including configurable Westgard rules, multi-level Levey-Jennings plots, and standard and reagent management of supplier details, lot numbers, and expiry dates. LabWare also has interfaces established to third-party quality control management systems, such as the Bio-Rad Interlaboratory Program and Randox Acusera.
LabWare’s plate management and Electronic Laboratory Notebook (ELN) modules work together seamlessly to streamline and automate the preparation and testing of multi-well plates for a variety of workflows (ELISA, DNA extraction, sequencing).
Reports are created according to the preferences of the recipient, the format/template for preliminary and final reports, units, delivery time, and mode of delivery.
The integrated Contact Manager module provides an automated phone queue for streamlined management and documentation of all follow-up communications to clinicians regarding orders and test results.
The Patient Manager module provides convenient access to all available laboratory test/result data, encounter details, charges and billing information, attached documents and files, clinical history, and demographic details, including family relationships.
The integrated Charge Manager module accumulates charges to invoices and accounts according to contracts and accounts defined for the submitting organization. Invoices can be reviewed, adjusted if necessary, printed, and closed as per the contract's requirements. Other features include volume discounting, managing local tax requirements, and multiple currencies for pricing and invoicing.
LabWare streamlines the effort involved with creating or modifying analytical methods with flexible test catalog setup capabilities, including:
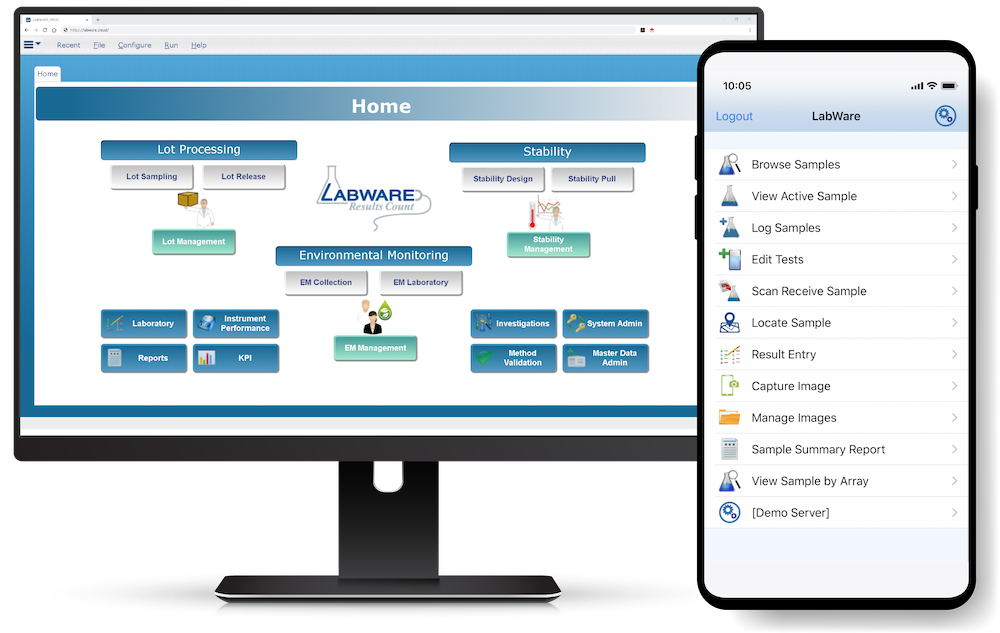
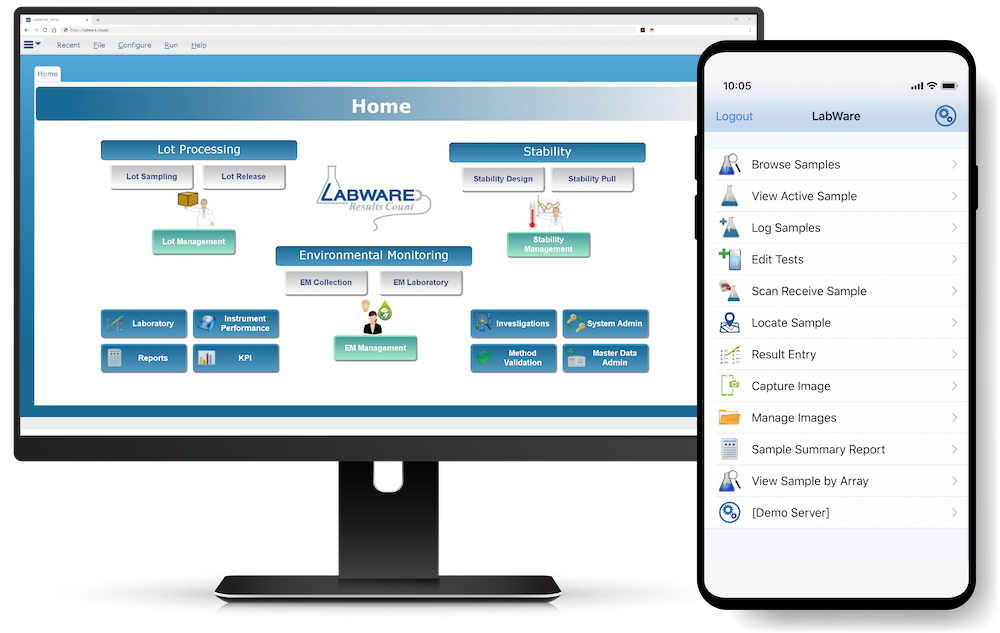
LabWare LIMS includes the following workflow management tools that help laboratories improve customer service, streamline laboratory operations, maintain regulatory compliance, and conduct clinical research.
LabWare can easily interface with other enterprise information systems such as Health Information Systems (HIS), Electronic Medical Records (EMR), Enterprise Resource Planning (ERP), and Customer Relationship Management (CRM) systems.
LabWare has native tools that use data-driven configuration to establish HL7, web services, file transfer, or direct database communications with other information systems.
LabWare includes robust interfacing tools to enable unidirectional or bidirectional communications with laboratory instruments, plate readers, liquid handling/robotics platforms, and laboratory instrument data analysis software.
LabWare's built-in ASTM communications module establishes unidirectional or bidirectional communications with clinical laboratory analyzers via ASTM or proprietary instrument-specific message formats.
LabWare’s integrated LabStation instrument interfacing tool can interface with various laboratory instruments via serial RS-232 communications (COM ports) or file-based import/export. LabStation parses the instrument output and automatically enters the results into LIMS, or results can be manually entered upon your review and approval.
LabWare also supports bidirectional communications with third-party instrument data acquisition tools, such as Data Innovations. It has a standard HL7 interface to the Data Innovations Instrument ManagerTM Software.
The following LabWare outreach tools support the delivery of customer service excellence:
LabWare’s Clinical Health Solution enables reference laboratories, hospitals, and other health care facilities to streamline the complete clinical order lifecycle for all medical laboratory disciplines, including biochemistry, hematology, microbiology, molecular/genomics, anatomic pathology, serology, immunology, virology, and urinalysis.
LabWare supports clinical laboratory automation and paperless operations with native tools that simplify interfacing with various laboratory analyzers and robotics platforms and a configurable rules engine to enable, expedite, and increase the efficiency and effectiveness of laboratory operations.
Key features include:

LabWare provides the most complete public health LIMS solution on the market. Our approach delivers a solution up front that includes all the necessary functionality required by a public health lab to enable workflow automation, increased efficiency and throughput, timely reporting & improved data exchange for disease surveillance, food safety, and monitoring for environmental and bio/chemical terror threats.
Key Features include:
LabWare's powerful laboratory workflow management tools help veterinary diagnostic and research labs realize benefits such as:
LabWare’s comprehensive molecular LIMS solution enables genomics and molecular pathology labs to efficiently manage a variety of frequently changing multi-step workflows, including nucleic acid extraction and quality testing, multiple PCR methodologies, library preparation, and QC, sequencing (NGS & Sanger), microarrays, flow cytometry, LC-MS, LC-MS/MS, karyotyping, FISH, and more.
LabWare’s workflow tools include:
• End-to-end specimen tracking, including genealogy, real-time workflow status monitoring, and location.
LabWare’s anatomic pathology solution is available as a robust standalone system or as part of a wholly-integrated clinical laboratory solution. Powerful workflow management tools include:
• Powerful reporting tools that enable the inclusion of annotated images in reports, convenient access to canned text phrases, the ability to produce a cross-lab discipline report, and editing capabilities similar to Microsoft Word.
LabWare's phlebotomy solution suits the needs of different order processing scenarios, such as routine ward collection rounds and outpatient clinics. Seamlessly integrate your phlebotomy operations with order processing. Phlebotomists are provided with comprehensive information about sample types, sample volumes, specimen collection tubes, storage conditions, and draw instructions automatically derived from the original test order.
Key features include:




→ 4 indicators that your lab is ready for a new LIMS
→ Breakdown of LIMS deployment options (SaaS v. PaaS v. Self-hosted)
→ 8 factors that determine the total cost of ownership of your LIMS


→ 5+ technical innovations built in LabWare 8
→ Visualizations of your instruments and systems interacting with LabWare 8
→ Overview of LabWare Mobile that brings portability to lab automation

LabWare is glad to announce its upcoming Nordic User Meeting, scheduled for 26–27 November 2025 at the Clarion...
Image source: Mettler Toledo. “LabX–LabWare Connector.” mt.com
Modern labs must operate more efficiently, deliver acc...
SaaS LIMS helps laboratories cut costs, maintain compliance, achieve scalability, and streamline inventory management....